I trained as a dentist back in the 1980s. I am happy that much of what I learned in dental school so long ago may no longer apply. In fact, recent research on the oral microbiome has led me to a new understanding of oral health and disease. These discoveries are changed my thinking about the role of the oral microbiome in systemic chronic disease. In particular, researchers are uncovering a previously hidden connection between oral diseases and autoimmune diseases.
Contrary to what I was taught, the goal of good oral hygiene is no longer just killing “bad” bacteria. Today, we are more focused on balancing the ecology of the oral microbiome. Case in point, on a recent trip to NYC I met with Dr. Gerry Curatola, who seems to agree with this approach in his recent book, The Mouth-Body Connection (1).
“Not only do we know that human health depends on maintaining balanced relationships…within complex communities of microorganisms, but microbes are recognized to contribute to disease in previously unexpected ways… overzealous oral hygiene is being recognized as a cause of disease.”
Curatola, 2017
The mouth is a gateway to the rest of the body
While the mouth is one of the most vital body parts to our health, we often overlook its significance. In fact, as I researched the oral microbiome I was surprised by many new facts about oral health. This was despite my background as a clinical dentist!
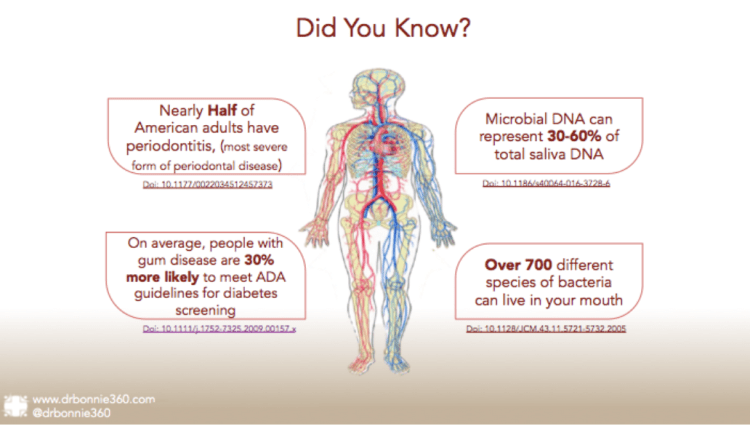
I was startled to find that nearly half of American adults have periodontitis. This is the most severe form of gum disease. But did you know that serious gum disease increases your likelihood of meeting ADA guidelines for diabetes screening? And by a shocking average of 30%?2 We cite more statistics below.
What is the oral microbiome?
Over the past 15 years, microbiologists have contributed hundreds of species to the Human Oral Microbiome Database(HOMD). Furthermore, they have cultured 68% of an estimated 700 different species of oral bacteria. To repeat, 700 different species of oral bacteria! That doesn’t include fungi or viruses, also key players in oral ecosystems. I could even have CosmosID profile my own oral microbiome populations.
The microbiome is highly variable both between and within individuals. Even more, various sectors of an individual’s microbiome show great diversity between compartments in the body. Species of bacteria present differ by location (e.g., hair, skin, stomach, and oral cavity, as shown below).
In the GI tract high levels of species diversity are correlated with better health. In contrast, oral disease is associated with an increased diversity and richness of the oral microbiome. As shown below, the oral cavity varies in species diversity. There are additional influences from biofilms and environmental conditions that may hinder or enhance disease.
Multiple human microbiomes
 Source: Frontiers in Microbiology
Source: Frontiers in Microbiology
Why should we care about our oral microbiome?
New understanding of the oral microbiome is shaping how we think about caries, periodontal and systemic diseases. The traditional view was that these diseases were caused by a limited number of pathogens. But now, we think of the oral microbiome as finely tuned communities. The balance of these communities influences not only oral health and disease, but also some systemic diseases (3). We describe an unbalanced microbiome with the term dysbiosis.
What happens when the oral microbiome is out of balance?
The mouth is the gateway between the outside world and our immune systems. Many things: food and drink, nutrients, microbes and toxins enter through the mouth. Moveover, the oral microbiome seeds the rest of the GI tract. There is a 45% overlap between the microbes found in the mouth and those in the colon.
The oral microbial ecosystem is vital to maintaining oral and systemic health. Saliva and biofilms on the teeth and soft tissue maintain balance in the oral cavity. This protects the mouth against pathogen overgrowth. In contrast, an unbalanced oral cavity ecosystem can stir pathogen activity and lead to oral disease.
A healthy oral microbiome needs good oral hygiene and a well functioning immune system. As shown below, poor oral hygiene, immune disorders and genetic predispositions all contribute to the disease cycle.
Multiple risk factors for oral dysbiosis & disease
A number of risk factors are shown below. Any of them may cause microbes to grow abnormally or become more virulent, consequently leading to disease. The key to oral health is maintaining balance among the diverse microbes of the oral microbiome. These microbes can interact both positively and negatively with the host. We do not yet have a good understanding of the cause and effect of ecological shifts in oral microbiome composition.

Source: Wiley Online Library
Decay is a polymicrobial disease, influenced by many factors
In dental decay (aka caries), carbohydrates ferment to organic acids (shown in red below). These organic acids lower local pH. Consequently, low pH demineralizes the tooth surface and drives dysbiosis in which both acid-producing and acid-tolerating microbiota are present. On the other hand, in a healthy oral cavity (in blue) shows a more complex pattern of metabolism. This pattern involves the catabolism (breakdown) of salivary proteins and glycoproteins. This process raises the pH of the mouth. Such a more alkaline mouth neutralizes microbial acid products.
However, dry mouth, too much snacking, smoking and other risk factors promote demineralization (net mineral loss). Eventually, demineralization leads to tooth decay, and caries or cavities.
Traditional caries-associated species include Streptococci mutans and Lactobacillus species. Increasing evidence points to the roles of Actinomyces, Bifidobacterium, and other Gram-positive rod species as well.
 Source: British Dental Journal
Source: British Dental Journal
The oral microbiome in periodontal health and disease
Recent advances in metagenomic, metatranscriptomic, and mechanistic studies suggest a new model of periodontal disease, as shown below. To explain, in this model, disease results from polymicrobial synergy and dysbiosis. It follows that persistent dysbiosis unbalances the mouth from homeostasis to disease. Furthermore, this imbalance affects not just the mouth but also distant sites in the body (6).

Source: US National Library of Medicine National Institutes of Health
Recent research suggests that potassium may be a key signal in host-microbiome dysbiosis in periodontitis. Moreover, the role of potassium may point to how changing diet could prevent periodontal disease progression (7).
The oral microbiome in immune health & autoimmune disease
New data suggests that changes in the interactions between the core (baseline) and variable microbiome are linked to several autoimmune diseases, as shown below.
Changes in the oral microbiome can be triggered by the host’s environment, genetic susceptibility, diet, smoking, etc. Thus, these factors may contribute to the pathogenesis of several diseases. Examples include Sjogren’s syndrome, rheumatoid arthritis (RA), systemic lupus erythematosus (SLE or lupus), and Crohn’s disease. (Read here to find out the microbiome’s role in these diseases.) Moreover, there is a delicate balance between the immune system and the microbiota. Therefore, disturbing that balance (dysbiosis or microbial imbalance) can trigger autoimmune progression or flares (8). This is a mechanism for how the oral microbiome influences health and disease.
By Bonnie Feldman, DDS, MBA and Ellen M. Martin
Interested in delving deeper into the role of the oral microbiome in autoimmune health? Explore this curated bibliography of recent research publications.
Click here for more autoimmune resources.
To learn and engage more… attend a session at the 5th Microbiome R&D and Business Collaboration Forum. See what it’s all about here!
———————————-
References
[1] Curatola, Gerald P. et al., “The Mouth-Body Connection: a 28-Day Program to Create a Healthy Mouth, Reduce Inflammation, and Prevent Disease throughout the Body,” Center Street, (2017). https://books.google.com/books/about/The_Mouth_Body_Connection.
[2] Strauss, S. M., et al., “The dental office visit as a potential opportunity for diabetes screening: an analysis using NHANES 2003-2004 data.” Journal of Public Health Dentistry (2010): 70: 156–162. http://onlinelibrary.wiley.com/doi/10.1111/j.1752-7325.2009.00157.x/full
[3] Zhang, Xuan et al. “The Oral and Gut Microbiomes Are Perturbed in Rheumatoid Arthritis and Partly Normalized after Treatment.” Nature Medicine 21.8 (2015): 895–905. http://www.nature.com/nm/journal/v21/n8/abs/nm.3914.html.
[4] Zarco, Vess et al. “The Oral Microbiome in Health and Disease and the Potential Impact on Personalized Dental Medicine.” Oral Diseases 18.2 (2012): 109–120. http://onlinelibrary.wiley.com/doi/10.1111/j.1601-0825.2011.01851.x/full.
[5] Dewhirst, Floyd E. “The Oral Microbiome: Critical for Understanding Oral Health and Disease.” Journal of the California Dental Association 44.7 (2016): 409–10. https://www.ncbi.nlm.nih.gov/pubmed/27514152.
[6] Hajishengallis, George. “Periodontitis: from microbial immune subversion to systemic inflammation,” Nature Reviews Immunology (2015): 30–44. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4276050/.
[7] Yost, Susan et al. “Potassium is a key signal in host-microbiome dysbiosis in periodontitis,” PLOS Pathogens (2017): e1006457. https://www.ncbi.nlm.nih.gov/pubmed/28632755.
[8] Nikitakis, N. G., et al. “The autoimmunity–oral microbiome connection.” Oral diseases (2016). http://onlinelibrary.wiley.com/doi/10.1111/odi.12589/full.






5 Comments
Gerry Curatola DDS · October 14, 2017 at 8:26 am
This is a terrific overview of the oral microbiome and offers a better understanding of the beneficial and essential role of the microbial community in our mouths that has long been misunderstood….and mistreated. It also sets the table for novel approaches in treatments and greater systemic health! Thank you Dr. Bonnie.
iSmile Orthodontics · September 13, 2018 at 12:24 pm
Great post, thanks for sharing!
Navigating Food in a World Where One Diet Does Not Fit All! – Your Autoimmunity Connection · July 20, 2018 at 8:52 pm
[…] Full of Bacteria; The Rising Culture of Probiotics, Pills, and Fruity Tea to Reduce Oral Pathogens; The Oral Microbiome: Critical for Understanding Oral Health and Disease; Hyperbiotics: The Mind behind the probiotics, for the Body, driven by […]
Dentalcidin: For Dirty Dentition – Your Autoimmunity Connection · November 21, 2019 at 10:14 pm
[…] not only get rid of harmful bacteria, but also helpful bacteria. This disrupts your natural microbiome and opens your mouth to other infections. So, while the S. mutans may be gone, an imbalanced […]
Bill Conlan & CrossBridge (Part 2) – Your Autoimmunity Connection · May 3, 2021 at 9:29 pm
[…] are modalities like exercise, diet, oral health and trigger avoidance that likely benefit patients. However, conventional clinical practices […]