Written by: Bonnie Feldman, DDS, MBA, Ellen M. Martin, Hailey Motooka, Sarah Hinton
Astonishingly, some 90% of Americans cannot name a single autoimmune disease. This is despite that 16% of the US population suffer from one or more such disorders—more people than cancer or cardiovascular disease. One source of confusion is that there are at least 80-100 autoimmune diseases, many rare. Individual disease names: Celiac, Crohn’s, Lupus, and Rheumatoid Arthritis may not be familiar, depending on our experience. To many, these words could pass as Pokemon names or Scrabble plays. To others, they are medical jargon in advertisements for new medications with scary side effects. One way or another, most of us have no understanding of what these words mean, even if you append the word disease to them. Likewise, similar problems across all immuno-inflammatory conditions makes this an invisible epidemic.

Why is Autoimmune Disease Unheard Of?
So, why are people so incognizant of these diseases and their large and growing prevalence?
Maybe we just know too little about what autoimmune diseases really are. For decades, practitioners and researchers classified autoimmune diseases by focusing on specific locations in the body. For example, RA is treated by rheumatologists because it causes joint pain and damage, while MS is treated by neurologists because the autoimmune attack is on nerve cells. In general, the definition of autoimmune disease is the immune system confusing normal cell components (antigens) with those of foreign or diseased cells or pathogens and thus mistakenly attacking its own healthy tissues. However, with continuous improvements in medical technologies, including data analytics, we are gaining a better understanding of the mechanisms that underlie autoimmunity. As a result, our understanding of autoimmune disease has evolved, along with our language to describe related, sometimes newer concepts.
Over the course of our research within the vast literature of autoimmunity, we have discovered discrepancies in how diseases are defined. After digging deeper, we found ourselves immersed in a sea of papers containing new classifications and vocabulary. Initially, the learning curve was steep, but over time, we were able to detangle some key terms and definitions. Discussing these may help researchers striving for answers, patients trying better understand their own disease, and the clinicians that diagnose and treat them.
Immune-Mediated Inflammatory Disease: A Novel Umbrella Term
Definition: Immune-mediated inflammatory disease (IMID) is “a concept used to collectively describe a group of ostensibly unrelated conditions that share common inflammatory pathways.”
This proposed nomenclature describes a spectrum of inflammatory diseases. This continuum of conditions can be classified as autoimmune, autoinflammatory or overlapping combinations. As defined, IMID encompasses two branching subcategories: autoinflammatory and autoimmune.
Innate and Adaptive Immune Systems
First, some background on the normal functioning of your body’s double immune systems, called Innate and Adaptive (or acquired).
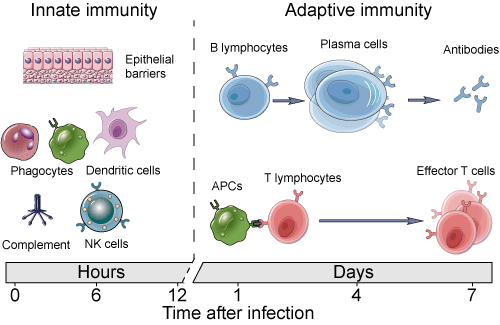
Innate immunity is the body’s first line of defense against foreign pathogens. They’re like nightclub bouncers, screening people before they enter, or removing them when they misbehave. However, instead of brute strength, their defense tactics consist of cellular and biochemical mechanisms that evolved to respond rapidly to infections. We inherited our innate immune systems from our evolutionary ancestors going back at least half a billion years. Therefore, we share many of these mechanisms with distant forms of life like jellyfish, molluscs and worms. Some mechanisms of innate immunity evolved to deal with molecules common to groups of related microbes (e.g., lipopolysaccharides (LPS) of gram-negative bacteria detected by Toll-like receptors). They do not distinguish finer differences between microbes. Other mechanisms are even more general, serving to prevent any pathogen from entering the body.
The principal components of innate immunity include:
- Physical and chemical barriers, such as skin, epithelial membranes and antimicrobial chemicals produced at epithelial surfaces, many by commensal microbes. These barriers don’t need to distinguish friend from foe, but like a castle’s moat or walls, keep random organisms or chemicals out of the body altogether.
- Phagocytic cells (neutrophils, macrophages), dendritic cells, natural killer (NK) cells and other innate lymphoid cells. These white blood cells sequester and consume pathogens or other foreign cells, as well as sending chemical signals to the innate and adaptive systems.
- Chemokines, blood-carried proteins, including members of the complement system and other mediators of inflammation. These chemicals trigger a cascade of effects, including local inflammation (e.g., redness and swelling at an infection site), general fever, and signaling to the rest of the immune system.
While many different types of cells are involved in the innate immune response, perhaps the most familiar are the macrophages. These large, mobile white blood cells cruise the vascular and lymphatic vessels to locate foreign bodies and essentially eat them, a process called phagocytosis. Moreover, upon detection of foreign viral or bacterial proteins, macrophages secrete signaling molecules called cytokines. Once in the blood, these little chemical messengers carry signals to other cells in the body, activating the adaptive immune system.
The Acquired or Adaptive Immune System
Adaptive Immunity, also known as the “acquired immune system,” is executed by specialized white blood cells called B and T cells. These cells begin as generalized stem cells in the bone marrow and differentiate into cells that attack specific pathogens or infected cells. B cells produce antibodies as their primary chemical weapons, whereas T cells produce cytokines that orchestrate destruction of infected cells. Both types of cell also clone themselves to produce populations pre-programmed to attack previously encountered pathogens.
The major aspect of adaptive immunity that differentiates it from innate immunity is the ability to detect specific molecules. To clarify, specificity means that each population of immune cells specializes in a single signaling molecule or marker. A particularly powerful result is that they respond quickly to repeated exposures to the same species of pathogen. This is known as memory. You are probably familiar with this concept from childhood diseases (which confer immunity to repeated infections). Based on this knowledge, we have developed vaccines, which “train” the acquired system to recognize pathogens through controlled exposure to live, killed or key molecular fragments of target pathogens.
There are two types of adaptive immune responses:
- Humoral Immunity – mediated by antibodies produced by B-cells
- Cell-mediated Immunity – mediated by T-cells and their cytokines
Based on infections, vaccines or even asymptomatic exposure, the adaptive immune system learns which pathogens to attack. Once exposed to a pathogen, such as the measles virus, the adaptive immune system can identify the same pathogen if exposed a second time. Then, when encountered again, memory B-cells quickly produce antibodies that circulate and attack these specific pathogens.
Autoimmunity vs. Autoinflammatory
Autoinflammatory Disease: Malfunction in the Innate Immune System
Definition: Autoinflammatory diseases (AIDs) refer to a group of rare and hereditary diseases characterized by recurrent inflammation in the absence of infection, without the unusual presence of antibodies or antigen-specific T-cells typical of autoimmune diseases.
Simply put, in autoinflammatory conditions the innate immune system reacts without known cause and without effective control. This means that white blood cells and inflammatory chemicals become over-reactive and poorly regulated. That is, the system does not calm down in the absence of any known trigger.
Some autoinflammatory diseases:
- Familial Mediterranean Fever (FMF)
- Neonatal Onset Multisystem Inflammatory Disease (NOMID)
- Tumor Necrosis Factor Receptor-Associated Periodic Syndrome (TRAPS)
- Deficiency of the Interleukin-1 Receptor Antagonist (DIRA)
- Behçet’s Disease
- Chronic Atypical Neutrophilic Dermatosis With Lipodystrophy and Elevated Temperature (CANDLE)
Autoimmune Disease: Malfunction in the Adaptive (Acquired) Immune System
Definition: Autoimmune disease refers to dysfunctions of the acquired immune system’s defense mechanisms.
Adaptive immunity appears in bony fish about 400MYO, and evolved further in our mammalian ancestors. Therefore, we share adaptive immune system elements with all vertebrates. Acquired immunity attacks pathogens by learning to recognize their molecular signatures. Therefore, specialized immune cells (B and T cells) respond more quickly to repeated exposures. In autoimmune diseases, something goes wrong with this process. These immune cells mistakenly learn to target proteins of the body’s own healthy cells, triggering a cascade of progressive inflammation and tissue damage.
Some examples of autoimmune diseases are:
- Celiac Disease
- Hashimoto’s Thyroiditis
- Type 1 Diabetes Mellitus
- Sjogren’s Syndrome
- Inflammatory Bowel Disease (IBD): Crohn’s Disease and Ulcerative Colitis
- Rheumatoid Arthritis
- Psoriasis
- Systemic Lupus Erythematosus (SLE or lupus)
Based on these definitions, the distinguishing factor between autoinflammatory and autoimmune diseases is the specificity or lack thereof in the immune system’s dysfunctional innate or adaptive response.
The Disease Continuum

Diseases can be classified based on innate/adaptive immunity responses due to the differences between the two systems. Classifying complex immunology diseases is somewhat arbitrary. Boundaries are ambiguous in terms of clinical, biological, pathological and genetic features, as suggested in the illustration.
A spectrum or continuum is a concept to help researchers, clinicians, drug developers and patients recognize that many diseases cannot be classified as solely autoinflammatory or autoimmune. Chronic, waxing and waning inflammation is a hallmark of all IMID diseases, regardless of where they fall on the spectrum. Distinguishing diseases based on specific immunological response may aid in developing more precisely targeted and personalized therapies in the future. Moreover, it can inform discovering diagnostic biomarkers to better match therapies to each patient.
At Autoimmune Connect, we are dedicated to adapting to the ever-changing science and understanding of immune–mediated diseases. While we continue to dive into the research, explore our resources below:
References
- Arango Duque, Guillermo and Albert Descoteaux. “Macrophage cytokines: involvement in immunity and infectious diseases” Frontiers in immunology vol. 5 491. 7 Oct. 2014, doi:10.3389/fimmu.2014.00491https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4188125/
- Grateau, Gilles, et al. “How Should We Approach Classification of Autoinflammatory Diseases?” Nature News, Nature Publishing Group, 9 July 2013, www.nature.com/articles/nrrheum.2013.101.
- “Innate vs adaptive immunity.” Khan Academy. Khan Academy. 24 Jan. 2019 <https://www.khanacademy.org/test-prep/mcat/biological-sciences-practice/biological-sciences-practice-tut/e/innate-vs-adaptive-immunity>.
- “Innate and Adaptive Immune Mechanisms.” Immunohistochemistry Guide – Creative Diagnostics, www.creative-diagnostics.com/innate-and-adaptive-immunity.htm.
- Janeway CA Jr, Travers P, Walport M, et al. Immunobiology: The Immune System in Health and Disease. 5th edition. New York: Garland Science; 2001. Autoimmune responses are directed against self antigens. https://www.ncbi.nlm.nih.gov/books/NBK27155/
- Kuek, Annabel et al. “Immune-mediated inflammatory diseases (IMIDs) and biologic therapy: a medical revolution” Postgraduate medical journal vol. 83,978 (2007): 251-60. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2600039/
- Martz, Lauren. “Why Autoimmune Diseases Should Be Redefined by Molecular Pathway.” BioCentury, 1 Nov. 2018, 7:50, www.biocentury.com/bc-innovations/targets-mechanisms/2018-11-01/why-autoimmune-diseases-should-be-redefined-molecular-.
- Mmassy, Bruno. “Adaptive Immunity.” LinkedIn SlideShare, 3 July 2011, www.slideshare.net/MMASSY/adaptive-immunity-8494348.
- Pathak S, McDermott MF, Savic S. Autoinflammatory diseases: update on classification diagnosis and management. Journal of Clinical Pathology 2017;70:1-8. https://jcp.bmj.com/content/70/1/1
- Peckham, Daniel, et al. “The Burgeoning Field of Innate Immune-Mediated Disease and Autoinflammation.” The Journal of Pathology, vol. 241, no. 2, 2016, pp. 123–139., doi:10.1002/path.4812. https://onlinelibrary.wiley.com/doi/full/10.1002/path.4812
- Brodin, Petter, et al. “Variation in the Human Immune System Is Largely Driven by Non-Heritable Influences.” Cell, vol. 160, no. 1-1, 15 Jan. 2015, pp. 37–47., doi:10.1016/j.cell.2014.12.020.






1 Comment
Homepage · July 22, 2019 at 6:30 pm
… [Trackback]
[…] Informations on that Topic: drbonnie360.com/2019/01/29/the-future-of-the-autoimmunity-continuum/ […]